Reader's Favourite
Business
New In Technology

Sponsored
Check Top Writers Review for the best essay writing services
Travel
Featured Reviews
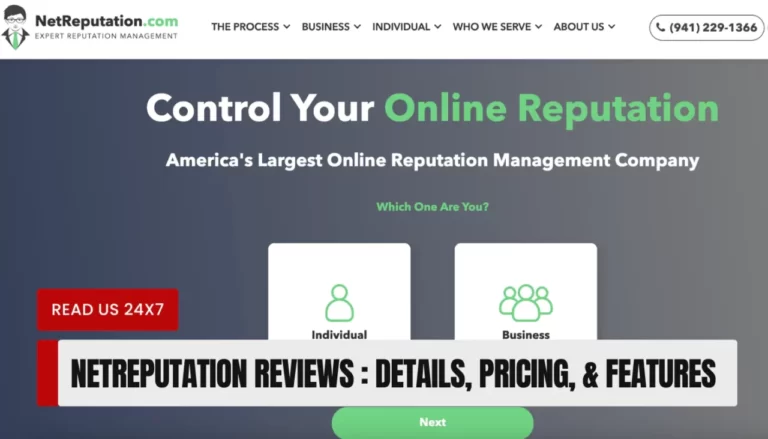
NetReputation is a legitimate and reputable online reputation management service that helps individuals and businesses establish, repair,…
Latest Articles
Many people mark May 1st on their calendars, but not everyone knows why. This date is Labour Day, also known as May…
The entertainment industry is mourning the loss of Scottish actor Brian McCardie. McCardie passed away unexpectedly at his home on April 28th,…
Feeling lost in the ever-evolving digital world? TrendzGuruji.me Awareness can be your guiding light. This platform empowers users to navigate the online…
Çeviit, a groundbreaking platform, has revolutionized the way we interact online. In this article, we delve into the definition of Çeviit and…
High-definition (HD) movies offer a superior viewing experience compared to standard definition. They provide sharper visuals, richer colors, and clearer sound, making…
VRChat login is your first step into a vast and exciting online world. It’s a free-to-play social platform where users can create,…
Craving spoilers for the heart-stopping romance “Fell into the Arms of a Mad Villain”? Look no further! This article dives into the…










































![How to Watch the 2024 Boston Marathon Online Today! [FREE] How to Watch the 2024 Boston Marathon Online Today](https://readus247.com/wp-content/uploads/2024/04/how-to-watch-the-2024-boston-marathon-online-661cfed4ef6e7-300x171.webp)


